$24M Verdict in Seattle Stem Cell Wrongful Death
A King County jury awarded $24 million after finding a Seattle stem cell clinic’s negligence caused a patient’s death following spinal injection.
Published on
A King County Superior Court jury awarded $24 million to the family of Michael “Mike” Trujillo, a 62-year-old Colorado electrician who died one day after undergoing a spinal stem cell procedure at a Seattle clinic. Trujillo, diagnosed with ALS in 2017, remained active and continued working full time, according to testimony describing his condition and daily life before seeking treatment. His widow, Carmen Trujillo, and their five adult children brought wrongful-death claims alleging that a procedure performed while he was taking blood-thinning medication led to catastrophic complications. The verdict allocates damages among the estate, Trujillo’s spouse, and each child, and the clinic’s former owner has indicated plans to appeal.
Alleged Marketing and Patient Decision-Making
The lawsuit centered in part on how Seattle Stem Cell Center, operated by entities including US Stemology, presented stem cell therapy to patients with serious conditions. According to the family’s allegations, Trujillo learned of the clinic through online marketing that promoted stem cell treatments for a wide range of chronic and life-altering diagnoses, including ALS. After a free consultation, he reportedly came to believe the therapy might improve his symptoms or offer meaningful hope of treating the disease, a central narrative at trial as jurors evaluated whether the care provided aligned with appropriate medical standards and disclosures.
The case unfolded against a broader regulatory backdrop concerning the marketing of stem cell interventions. Public statements in separate state enforcement litigation had asserted that the clinic treated more than 100 patients for serious conditions and emphasized that reliable clinical evidence did not support claims that stem cell therapy could effectively treat many of those conditions. While the wrongful-death action focused on clinical decision-making and informed consent issues specific to Trujillo’s care, the context reinforced disputes common in these cases: the line between promotional representations and scientifically supported outcomes, and whether vulnerable patients facing progressive illness are given balanced information about risks, benefits, and alternatives.
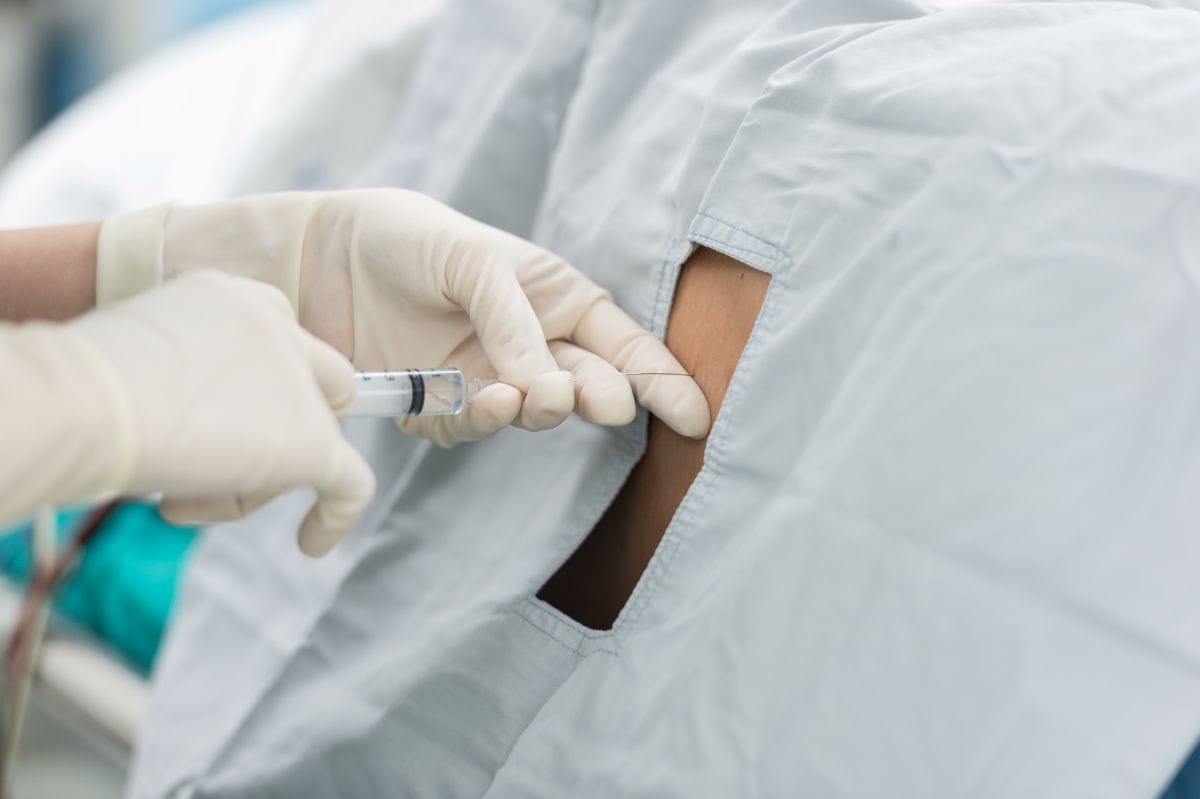
Treatment Timeline and Negligence Findings
Evidence at trial addressed two procedures in 2019, including a second treatment in early April when stem cells were injected into Trujillo’s spine. According to the complaint, Trujillo was taking Coumadin, a blood-thinning medication, and he informed the clinic. The family alleged he was never instructed to stop the anticoagulant before either procedure. Trial evidence, as described in accounts of the proceedings, indicated that the spinal procedure was performed without imaging guidance while Trujillo was on blood thinners, and that this combination contributed to severe bleeding and subsequent brain herniation. The complaint similarly alleged that the spinal injection caused significant brain bleeding, and Trujillo died the following day.
The jury returned a unanimous verdict and concluded that negligence by clinicians involved in his treatment caused the fatal outcome, according to summaries of the decision. Damages totaled $24 million, including $4 million to Trujillo’s estate, $5 million to Carmen Trujillo, and $3 million to each of the couple’s five children. In allocating these amounts, the verdict reflected both economic and noneconomic harms typically evaluated in wrongful-death and survivorship claims, including loss of companionship and the decedent’s pre-death suffering as presented through medical evidence and family testimony.
Post-Verdict Positions and Regulatory Context
Following the verdict, the clinic’s former owner, Dr. Tami Meraglia, stated that Seattle Stem Cell Center closed in 2021 and that she intends to appeal. She asserted that pretrial and trial rulings limited what the defense could present, stating, “Based on legal rulings made before and during the trial, the jury did not hear all of the evidence we sought to present.” She also maintained that she did not personally see Trujillo and that another physician performed the procedure. In her account, the plan changed from an IV approach to an epidural spinal procedure, and she characterized the change—while the patient was on a blood thinner and had high blood pressure—as central to the alleged errors.
The verdict also arrived after separate consumer-protection litigation by Washington’s attorney general against US Stemology and Dr. Meraglia resulted in a consent decree that imposed permanent marketing restrictions and an $800,000 judgment. That matter, while distinct from the wrongful-death claims, underscores how marketing, clinical practice, and regulatory oversight can converge when clinics offer stem cell interventions outside FDA-approved indications. Publicly stated federal guidance has emphasized that FDA-approved stem cell treatments are limited to certain blood disorders and involve different cell types than those used in many commercial stem cell offerings; courts and regulators often scrutinize how clinics communicate that distinction to patients making high-stakes medical decisions.
Case Details
Case Name: Trujillo et al. v. US Stemology LLC et al.
Court Name: King County Superior Court
Case Number: 22-2-00729-0
Plaintiff Attorney(s): Swanson Gardner Meyers Cohon